Preclinical Study
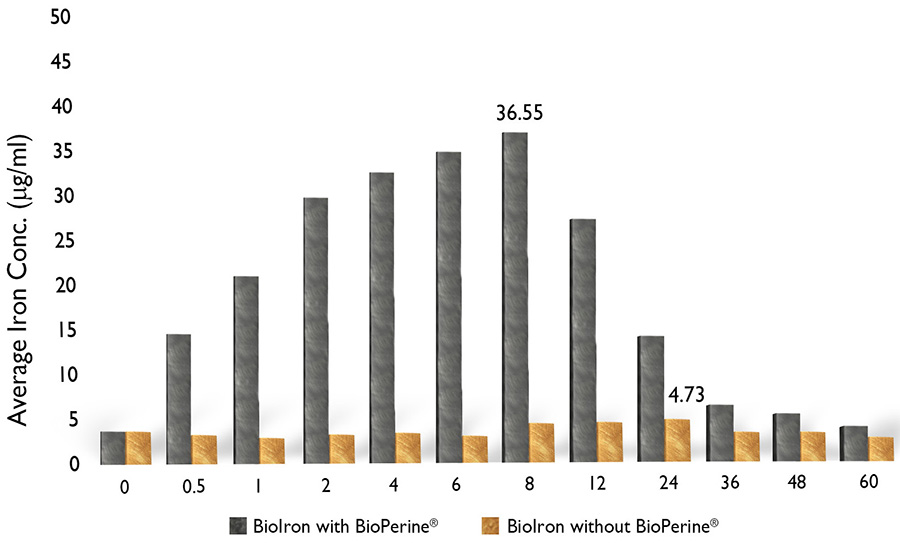
An in vivo study was performed to establish and validate the bioavailability of organic elemental iron (BioIron) in combination with or without BioPerine®.
The study involved a single oral dose of BioIron with BioPerine® (group I) or BioIron without BioPerine® (group II) administration to rabbits, and blood samples were collected at different time points.
Results showed that at 8 h time point, serum concentration of iron was significantly higher in group treated with BioIron containing BioPerine® (36.55 ± 9.97 μg/ml) when compared to group treated with BioIron containing no BioPerine®.